|
sintofen
Plant growth regulator
NOMENCLATURE
Common name sintofen (pa ISO)
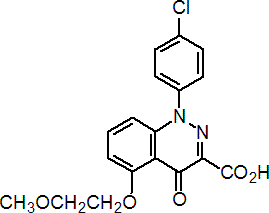
IUPAC name 1-(4-chlorophenyl)-1,4-dihydro-5-(2-methoxyethoxy)-4-oxocinnoline-3-carboxylic acid
Chemical Abstracts name 1-(4-chlorophenyl)-1,4-dihydro-5-(2-methoxyethoxy)-4-oxo-3-cinnolinecarboxylic acid
CAS RN [130561-48-7] Development codes SC 2053 (Orsan)
PHYSICAL CHEMISTRY
Composition Tech. is 99.7%. Mol. wt. 374.8 M.f. C18H15ClN2O5 Form Cream to yellow powder. M.p. 261.03 °C V.p. 1.1 ´ 10-3 mPa (25 °C, gas saturation method) KOW logP = 1.44?.06 (25? °C) Henry 7.49 ´ 10-5 Pa m3 mol-1 S.g./density 1.461 (tech., 20 °C) Solubility In water <5 mg/l (20 °C). In methanol, acetone and toluene <0.005, 1,2-dichloroethane 0.01-0.1 (all g/l, 20°C). Stability Stable in aqueous solution; DT50 >365 d (50 °C, pH 5, 7 and 9). pKa 7.60
APPLICATIONS
Mode of action Cereal hybridising agent. Uses Hybridisation agent used at 0.8 kg/ha in France for sterilising spring and winter wheat (Triticum aestivum). Formulation types SL. Selected products: 'Croisor' (DuPont)
MAMMALIAN TOXICOLOGY
Oral Acute oral LD50 for rats >5000 mg/kg. Skin and eye Acute percutaneous LD50 for rats >2000 mg/kg. Inhalation LC50 (4 h) for rats >7.34 mg/l. NOEL (2 y) for rats 12.6 mg/kg b.w. daily, expressed as the salt. ADI 0.126 mg/kg b.w.
ECOTOXICOLOGY
Birds Acute oral LD50 for mallard and bobwhite quail >2000 mg/kg. LC50 (8 d) for bobwhite quail >5000 ppm. Fish LC50 (96 h) for rainbow trout 793, bluegill sunfish 1162 mg/l. Daphnia EC50 (48 h) 331 mg/l. Algae EC50 (96 h) for Selenastrum capricornutum 11.4 mg/l. Bees LD50 (48 h) oral and contact >100 µg/bee. Worms LC50 (14 d) for Eisenia foetida >1000 ppm.
ENVIRONMENTAL FATE
Animals Rapidly excreted in mammals, primarily via the urine. Plants Metabolism studies in wheat support the residue definition of sintofen plus metabolite SC 3095 (1-(4'-chlorophenyl)-1,4-dihydro-5-(2"-hydroxyethoxy)-4-oxo-cinnoline-3-carboxylic acid). Soil/Environment Degraded slowly in soil, DT50 (lab.) 130-329 d (20°C, 40-50% MHC). Not prone to leaching; Koc 376-18 848 (soil % o.c. 0.99-10.92). Stable to hydrolysis at pH 5, 7 and 9. In non-sterile water sediment systems (lab., 20°C) it rapidly disappears from water; DT50 6.7-20.1 d; the main route of dissipation is partitioning to sediment; DT50 in the total system >105 d.
|