|
cinosulfuron
Herbicide
HRAC B WSSA 2; sulfonylurea
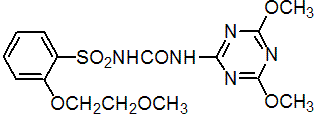
NOMENCLATURE
Common name cinosulfuron (BSI, draft E-ISO)
IUPAC name 1-(4,6-dimethoxy-1,3,5-triazin-2-yl)-3-[2-(2-methoxyethoxy)phenylsulfonyl]urea
Chemical Abstracts name N-[[(4,6-dimethoxy-1,3,5-triazin-2-yl)amino]carbonyl]-2-(2-methoxyethoxy)benzenesulfonamide
CAS RN [94593-91-6] Development codes CGA 142 464 (Ciba-Geigy)
PHYSICAL CHEMISTRY
Mol. wt. 413.4 M.f. C15H19N5O7S Form Colourless crystalline powder. M.p. 127.0-135.2 ºC (pure) V.p. <0.01 mPa (25 ºC) (OECD 104) KOW logP = 2.04 (pH 2.1, 25 ºC) Henry <1 ´ 10-6 Pa m3 mol-1 (pH 6.7, calc.) S.g./density 1.47 (20 ºC). Solubility In water 120 (pH 5.0), 4000 (pH 6.7), 19 000 (pH 8.1) (all in mg/l, 25 ºC). In acetone 36 000, ethanol 1900, toluene 540, n-octanol 260, n-hexane <1 (all in mg/l, 25 ºC). Stability Decomposes above the melting point. No significant hydrolysis at pH 7-10; considerable hydrolysis at pH 3-5. pKa 4.72
COMMERCIALISATION
History Herbicide reported by M. Quadranti et al. (Proc. 11th Conf. Asian Pacific Weed Sci. Soc., 1987, 1, 117). Introduced by Ciba-Geigy AG (now Syngenta AG). Patents US 4479821; EP 44807 Manufacturers Syngenta
APPLICATIONS
Biochemistry Branched chain amino acid synthesis (ALS or AHAS) inhibitor. Acts by inhibiting biosynthesis of the essential amino acids valine and isoleucine, hence stopping cell division and plant growth. Selectivity derives from rapid metabolism in the crop. Metabolic basis of selectivity in sulfonylureas reviewed (M. K. Koeppe & H. M. Brown, Agro-Food-Industry, 6, 9-14 (1995)). Mode of action Absorbed primarily by shoots and roots, and translocated to actively growing meristematic tissue. Uses Applied post-emergence to control many weeds, including Alisma, annual Cyperus, Eleocharis, Marsilea, Potamogeton and Sagittaria spp., Monochoria vaginalis and Sphenoclea zeylanica in transplanted, direct seeded, wet-sown and water-sown, and dry-sown flooded rice crops, at 20-80 g/ha. Also used in tropical plantation crops. For full weed spectrum, may need to be combined with a grass herbicide in tank-mix or sequential treatment. Phytotoxicity No varietal restrictions for rice. Formulation types GR; WG. Selected products: 'Setoff' (Syngenta); mixtures: 'Apiro Ace' (+ pyriftalid) (Syngenta)
OTHER PRODUCTS
Mixtures: 'PIPSET' (+ piperophos) (Syngenta) Discontinued products: 'Sailant' * (Ciba); 'Sofit Super' * (Ciba)
ANALYSIS
Product and residue analysis by hplc. Methods for sulfonylurea residues in crops, soil and water reviewed (A. C. Barefoot et al., Proc. Br. Crop Prot. Conf. - Weeds, 1995, 2, 707). Details available from Syngenta.
MAMMALIAN TOXICOLOGY
Oral Acute oral LD50 for rats and mice >5000 mg/kg. Skin and eye Acute percutaneous LD50 for rats >2000 mg/kg; not irritant to skin or eyes of rabbits. Non-sensitising to skin (guinea pigs). Inhalation LC50 (4 h) for rats >5 mg/l air. NOEL (2 y) for rats 400, mice 60 ppm; (1 y) for dogs 2500 ppm. Toxicity class WHO (a.i.) U
ECOTOXICOLOGY
Birds Acute oral LD50 for Japanese quail >2000 mg/kg. Fish LC50 (96 h) for rainbow trout >100 mg/l. Daphnia LC50 (48 h) 2500 mg/l. Algae EC50 (72 h) for Scenedesmus subspicatus 4.8 mg/l. Bees Non-toxic; LD50 (oral and contact) >100 mg/bee. Worms LC50 (14 d) for Eisenia foetida 1000 mg/kg.
ENVIRONMENTAL FATE
Animals Hydrolysis of methoxy groups and cleavage of the sulfonylurea bridge. It is rapidly excreted (80-100% within 24 h). Plants In plants, rapid degradation by cleavage of the sulfonylurea bridge. Soil/Environment In soil, degrades (a) by O-dealkylation in both ring systems, forming the corresponding phenol, mono- and di- hydroxytriazine and (b) by cleavage of the sulfonylurea bridge; degradation proceeds further to form bound residues and CO2; DT50 in soil (lab.) c. 20 d, in paddy fields c. 3 d. No potential for bioaccumulation. Koc c. 20, indicating that cinosulfuron may leach; column data show however that rapid degradation prevents leaching. In paddy fields under natural conditions, cinosulfuron dissipates rapidly due to photolysis and, to a lesser extent, by adsorption to soil.
|